Defining Acid Rain - What It Is And Its Impact On The Environment
Defining Acid Rain - What It Is And Its Impact On The Environment

Pollutants like sulfur dioxide and nitrogen oxides can reach dangerously high levels in the atmosphere. Combined with water droplets and oxygen, they result in a phenomenon called acid rain.
If the conditions are favorable, a chemical reaction takes place between these harmful gases and natural elements like water and moisture in the air, resulting in a type of precipitation that is very harmful to both organic and inorganic matter on the surface.
This phenomenon is called acid rain. The term may sound ominous and invoke images of pure acid falling from the sky and eating away at anything it touches. Although the reality is not nearly as horrific, over time, acid rain still causes significant damage to the environment.
This article examines what acid rain is and how it forms. It then focuses on the important facts you need to know about this phenomenon.
What Is Acid Rain?
Before we take a closer look at how acid rain forms and its impact on the environment, we need a clear definition of what exactly acid rain is:
Acid Rain Definition
Acid rain is the umbrella term for any form of precipitation with unusually high acidity levels resulting from sulfur dioxide and nitrogen oxide emissions in the atmosphere. Combined with water, oxygen, and other chemicals, they form sulfuric and nitric acids that mix with moisture in the atmosphere to form acid rain.
Acid rain (also known as acid deposition) describes the different types of acidic particles that get deposited on objects on the planet’s surface.
Although the term may sound as if it refers to a form of “rain,” it implies all types of precipitation that contain unusually high acidity levels. It includes forms of precipitation like rainfall, hail, mist, and snow.
It is important to note that acid deposition does not only occur in liquid form. It can be categorized into two types. The first is quite familiar, while the second takes place almost unnoticed, potentially causing more damage. They are:
1) Wet Depositions
Acid rain in the form of wet depositions is the most familiar kind to observers. It includes any type of wet, acidic precipitation, like rain, snow, mist, and hail.
In regions with a high percentage of rainfall, this is the most common type of acidic deposition, as sulfuric and nitric acid bind with water in the atmosphere before falling to the ground.
2) Dry Depositions
Regions with very little or no rainfall are not exempt from acidic depositions. In countries like Egypt and other arid regions, acid deposition can occur in the absence of rain.
Here, acidic particles and gases attach directly to surfaces on the ground, including artificial structures, vegetation, and water bodies.
 For example, the effect of dry acid depositions on the famous pyramids of Giza has become a real and visible concern for archaeological conservationists in the region.
For example, the effect of dry acid depositions on the famous pyramids of Giza has become a real and visible concern for archaeological conservationists in the region.
When rainfall does occur, water can wash the acid away from the surface and carry it to lakes, dams, and other bodies of water, raising their level of acidity, which can severely affect aquatic life.
Depending on the absence of rainfall, dry acid deposition can account for as much as 20 – 60 percent of the total amount of acid deposition in a specific region.
To understand acid rain, one needs to understand acidity. Acidity is measured on the pH scale, which ranges from 0 to 14. The most acidic part of the scale is 0, the most alkaline 14, while 7 is the neutral value where an element is neither acidic nor alkaline.
Natural rainwater has a pH level of approximately 5.6 (due to the carbon dioxide present in rainwater, which makes it slightly acidic). Acid rain, however, has a pH level of 4.2 – 4.4 as a result of the presence of sulfuric or nitric acid in rainwater.
Although acid rain can cause long-term damage to the environment and structures, it is not nearly strong enough to cause immediate harm to humans. (For example, vinegar has a pH level of around 2.2 and is still safe for human consumption in moderate amounts.)
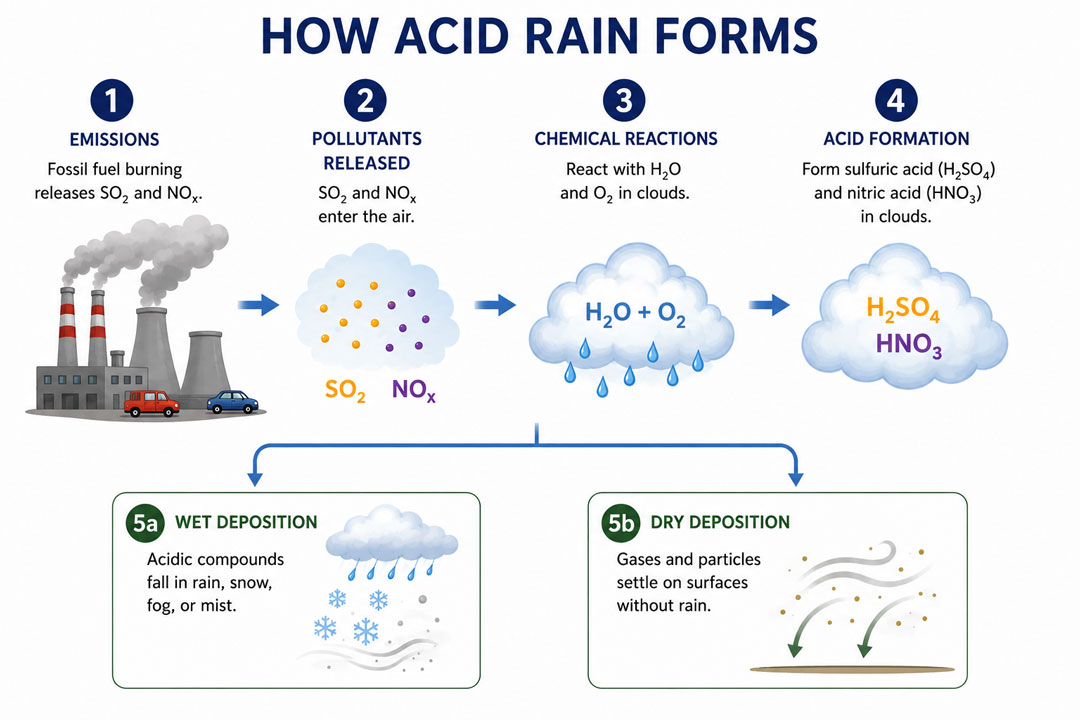
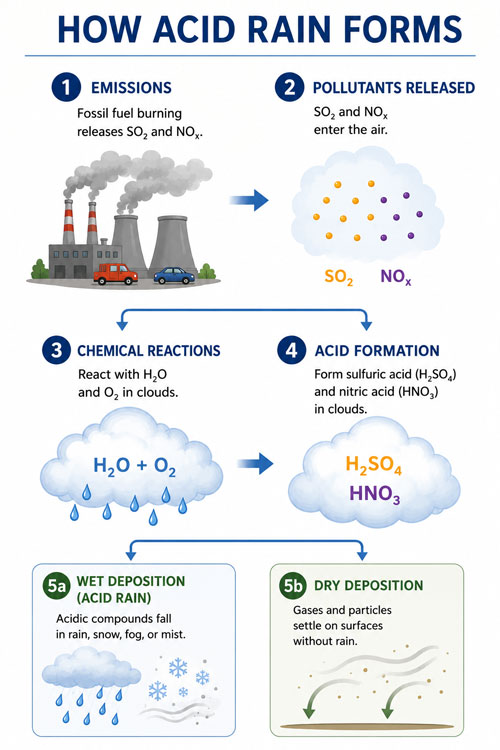
What Causes Acid Rain?
Click on the image for a larger view.
As mentioned in the previous section, rainwater already has a small level of acidity. It is not strong enough to cause any damage to the environment, though. However, when high levels of sulfur dioxide and nitrogen oxides are present in the atmosphere, they can result in acid rain.
Sulfur dioxide and nitrogen oxides do occur naturally. They are produced by volcanic eruptions and weather phenomena like lightning, and can also be the result of biological processes. But natural processes play a very small part in sulfur dioxide and nitrogen oxide production.
The vast majority of sulfur dioxide and nitrogen oxides present in the atmosphere are the result of the burning of fossil fuels (coal, natural gas, and oil) by humans.
 The burning of coal to produce electricity is the biggest culprit, closely followed by the emissions from automobiles and heavy industries.
The burning of coal to produce electricity is the biggest culprit, closely followed by the emissions from automobiles and heavy industries.
These sources release sulfur dioxide (SO₂) and nitrogen oxides (NOₓ) into the atmosphere. Once in the air, SO₂ and NOₓ react with water, various chemicals, and oxygen to form sulfuric and nitric acid.
Both sulfuric and nitric acid can combine with water and fall to the surface in the form of wet deposition. In arid regions, acidic gases and particles can also attach directly to objects on the ground in the form of dry deposition.
Acid rain does not only occur in regions where the toxic gases originate, as global winds can carry them over vast distances, resulting in acid rain on a different continent.
For example, late in the 20th century, most of the acid deposition in Norway originated in the United Kingdom, from where winds carried it over 900 km (560 miles), causing acid rain to destroy the trout and salmon population in Norwegian lakes.
Effects Of Acid Rain
It is clear that acid rain poses a significant threat to the environment. The biggest impact is on objects on the ground that are exposed to acid deposition over an extended period.
A wide range of organic and artificial objects are adversely affected by acid rain. They can be categorized into three sections:
-
- Effect On Bodies Of Water
- Damage To Vegetation
- Damage To Structures
To get a better understanding of how each category is affected by acid rain, one needs to examine each one in closer detail:
1) Effect On Bodies Of Water
Acid rain has an adverse effect on rivers, dams, and lakes. The most significant impact is on standing bodies of water, especially lakes, where acidity from various sources builds up in the water.
 Acid buildup in lakes is a result of direct precipitation, which is further accelerated by rivers carrying acidic water into the lakes. A further contribution comes from acid on the surface that gets washed away by rainwater and carried to lakes by overland flow.
Acid buildup in lakes is a result of direct precipitation, which is further accelerated by rivers carrying acidic water into the lakes. A further contribution comes from acid on the surface that gets washed away by rainwater and carried to lakes by overland flow.
This acid buildup has a deadly effect on aquatic life forms, including fish, shellfish, and even insects. It has already resulted in the complete elimination of some fish species, such as the brook trout, in some lakes.
(Earlier in this post, we already highlighted the deadly effect of acid rain on Norwegian lakes, where trout and salmon were almost completely wiped out from some lakes in the region.)
2) Damage To Vegetation
Acid rain has a severe effect on vegetation, especially tree life, in regions that experience high levels of acid deposition.
Although plant life and crops can also be affected, it is easier to control acidity in agricultural fields by adding fertilizer to replenish nutrients and using limestone to counter the acidity in the soil.
Trees are affected by the acid content in rainwater in different ways:
-
- Acidic compounds can strip away the protective layers on tree leaves, leaving the damaged leaves unable to photosynthesize.
- Acid rain can also infiltrate the soil around and below a tree, depleting the minerals and nutrients needed for growth.
- Finally, acid deposition can also release elements such as aluminum that are toxic to trees and other forms of vegetation.
The damage can be so severe that acid rain can result in large-scale tree death within a specific region. The Black Triangle on the German border is a region where the forests in the Jizera Mountains experienced widespread tree death due to heavy industrial activity.
3) Damage To Structures
Buildings, statues, and other artificially created structures are all affected by weathering over time. Acid rain, though, can significantly speed up the breakdown of some structures. Certain natural stone structures are also vulnerable to the effects of acid rain.
 Some materials are especially vulnerable to acid deposition and can deteriorate at an accelerated pace when subjected to acid rain. These materials include sandstone, limestone, marble, and a variety of metals.
Some materials are especially vulnerable to acid deposition and can deteriorate at an accelerated pace when subjected to acid rain. These materials include sandstone, limestone, marble, and a variety of metals.
Objects consisting of these materials can suffer severe deterioration or corrosion over time when they are exposed to acid rain. Gravestones and statues are notable examples of objects that can incur extensive damage due to acid deposition.
Acid Rain Facts
The best way to highlight the characteristics and importance of acid rain is to focus on some key facts about this phenomenon, which is also the subject of this article.
Some of the following points were already mentioned earlier in this post, but this list serves to highlight the essential facts and add a few new ones to give you a proper overview of what you need to know about acid rain:
-
- Acid rain does not pose any short-term danger to humans since the acid content is too diluted and weak to cause any harm.
- Long-term exposure to high levels of sulfuric and nitric acid in acid deposition can adversely affect people with underlying respiratory and cardiovascular conditions like asthma and heart disease.
- Acid rain can destroy aquatic life in bodies of water, like dams and lakes, by raising acidity levels in the water that make it uninhabitable.
- Acid deposition can adversely affect vegetation and destroy large sections of trees by depleting essential nutrients from the soil and removing the protective coating from leaves.
- Artificial structures, specifically those made of materials like limestone, marble, sandstone, and certain metals, are especially prone to damage and deterioration by acid rain.
- Acid deposition is not limited to regions where sulfur dioxide and nitrogen oxides originate, as global winds can blow the gases for hundreds of miles and cause acid rain in an entirely different location.
- Acidity is measured on the pH scale, ranging from 0 to 14, with 0 being the most acidic and 14 the most alkaline. Acid rain has a pH level of between 4.2 and 4.4.
- Acid rain can only be eliminated by dramatically limiting or halting the production of pollutants like sulfur dioxide and nitrogen oxides.
- Acid deposition does not only occur as a result of precipitation. In arid regions with little rain, dry deposition still occurs in the form of acidic gases or dust settling on surface objects.
- The phenomenon was discovered as early as the 17th century, and the term “acid rain” was coined by Robert Angus Smith in 1872.
- The gases responsible for the formation of acid rain also occur naturally in the atmosphere and are produced by processes like volcanic eruptions and lightning.
- The effects of acid rain can be combated. In agriculture, the effects of acid rain on vegetation are neutralized by adding materials like limestone and fertilizer to replenish depleted nutrients.
- Natural rainwater is also slightly acidic, with a pH level of 5.6 (with seven being neutral). It is due to the presence of carbonic acid in the water. This is still too weak to cause any damage to the environment, though.
- Acid rain has no smell. Unlike strong acids we are familiar with, acid rain is too diluted to have any potent smell. It smells no different than natural rainwater.
This is a summary of some of the key facts about acid rain. There is much more information available about this phenomenon, but this article captures the most relevant information and explanations.
Conclusion
Acid rain sounds scary, and although it poses little immediate danger to humans, the long-term effect on the environment can be quite severe and may harm or even destroy parts of the ecosystem.
Since acid deposition takes such a long time to show damage to objects, it was not that easy to provide adequate protection in time, if possible at all. Fortunately, the cause is now known, and governments have implemented regulations to limit the production of acidic gases.
In this article, we defined what acid rain is, how it is formed, and its impact on the environment. We concluded by highlighting some key facts about this phenomenon.
Until next time, keep your eye on the weather!